
Proper controls are essential for assessing signal specificity. Negative controls ensure that the observed signals are due to the presence of the target antigen and not due to non-specific binding or other artifacts. SYSY Antibodies recommends the following control experiments to ensure staining specificity.
Negative controls omitting the primary antibody help to identify false positive signals caused by the secondary system.
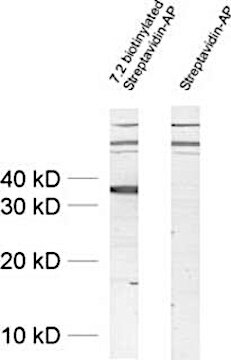
Figure 1: Detection of Synaptophysin with a biotinylated primary antibody (cat. no. 101 011BT) leads to the detection of unexpected high molecular weight signals. Omitting the primary antibody reveals that these signals originate from the secondary reagent streptavidin-AP.
Figure 1: Detection of Synaptophysin with a biotinylated primary antibody (cat. no. 101 011BT) leads to the detection of unexpected high molecular weight signals. Omitting the primary antibody reveals that these signals originate from the secondary reagent streptavidin-AP.
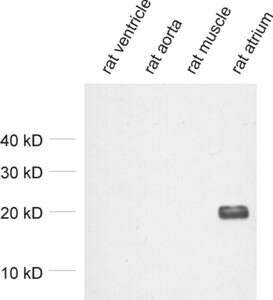
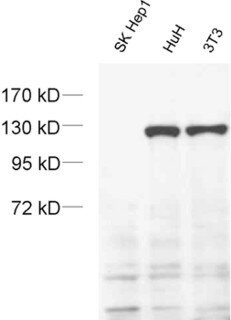
Figure 2: Detection of MLC-2A in tissue lysates from rat heart ventricle (negative), rat aorta (negative), rat skeletal muscle (negative) and rat atrium (positive) with mouse anti-MLC-2A antibody (cat. no. 311 011) confirming its specificity.
Figure 3: Detection of exportin4 in cell lysates from SK-Hep1 cells (negative), HuH cells (positive) and NIH 3T3 cells (positive) with mouse anti-Exportin4 antibody (cat. no. 215 011) confirming its specificity.
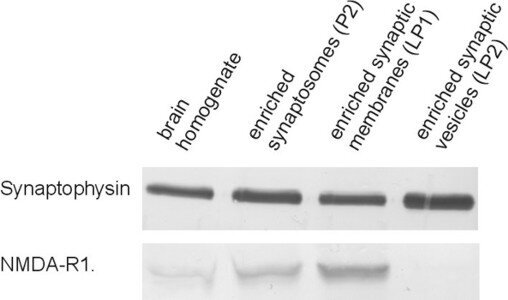
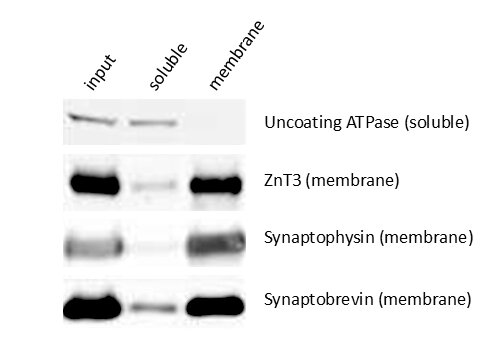
Figure 4: Detection of Synaptophysin and NMDA-R1 (GluN1) in different brain fractions showing the enrichment of GluN1 in synaptic membrane and its absence from synaptic vesicles. Synaptophysin is also present in the synaptic membrane fraction (LP1) due to fusion events of synaptic vesicles with the pre-synaptic membrane and is significantly enriched in the synaptic vesicle fraction LP2.
Figure 5: Detection of soluble (uncoating ATPase) and membrane bound proteins (ZnT3, Synaptophysin and Synaptobrevin) in the total input and soluble (aqueous) and membrane fractions of brain lysate. Integral membrane proteins are strongly enriched in the membrane fraction whereas water-soluble proteins can be detected in the aqueous phase.
To verify whether an antibody truly recognizes the intended protein - or cross‑reacts with closely related isoforms - a transiently over‑expression of the target and potential homologues in a eukaryotic cell line can be a useful approach. This strategy is especially valuable when several isoforms share high sequence homology. By fusing a short epitope tag (e.g., HA, ALFA, or FLAG) to each construct, the expression levels of the transfected proteins can be directly compared.
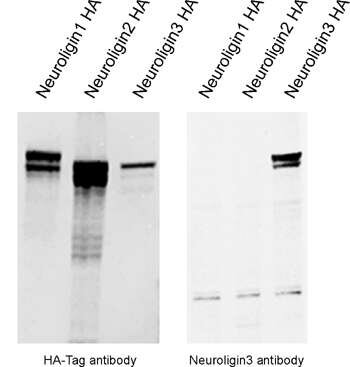
Figure 6: Overexpression of HA-tagged Neuroligin1, Neuroligin2 and Neuroligin3 confirms successful transfection of all three constructs (HA-Tag detection) and the specificity of the rabbit anti-Neuroligin3 antibody (cat. no. 129 113).
Figure 6: Overexpression of HA-tagged Neuroligin1, Neuroligin2 and Neuroligin3 confirms successful transfection of all three constructs (HA-Tag detection) and the specificity of the rabbit anti-Neuroligin3 antibody (cat. no. 129 113).