
The primary antibody is labeled with enzymes or fluorochromes, enabling immediate visualization without a secondary antibody. This method is fast and straightforward but may produce weak signals, especially when antigen expression is low.
A secondary antibody directed against the host species of the primary antibody amplifies the signal and is conjugated to a reporter, such as an enzyme (HRP or AP) or a fluorophore (Figure 1). An exception are biotin-labelled primary antibodies, which are detected with streptavidin or avidin conjugated to a reporter enzyme or fluorophore. Indirect detection methods amplify the signal and are generally preferred due to their higher sensitivity compared to direct labelling.
Antibodies from SYSY Antibodies are designed to deliver consistent and reliable detection, especially when used in accordance with our validated reference protocols.
Monoclonal antibodies are produced from a single clone of B cells (often fused with myeloma cells to create hybridomas) and consist of identical immunoglobulins, usually IgG or sometimes IgM. They recognize one specific epitope, providing high specificity with minimal cross-reactivity.
Recombinant antibodies are monoclonal antibodies that are produced in vitro by using known antibody coding genes. As they are produced under highly defined and controlled conditions they ensure exceptional batch-to-batch consistency (see also our Featured Topic on Recombinant antibodies).
Polyclonal antibodies are a mixture of antibodies produced by different B cell clones, each recognizing multiple epitopes on the target antigen. As a result, they are often more sensitive and tolerant to minor changes in antigen structure, but less specific than monoclonal antibodies, which can sometimes lead to increased background.
Although SYSY Antibodies provides recommended starting dilutions for all antibodies, empirical optimization is strongly advised, as the ideal dilution can vary depending on sample type and experimental conditions. For polyclonal antibodies derived from new immunizations, re-titration may be necessary to maintain optimal performance (see our FAQ on Synaptic Systems lot numbers for details.)
Basically, antibodies can be incubated at room temperature for a couple of hours with good results. Incubating primary antibodies overnight at 4 °C often enhances specificity and reduces background, making it particularly useful for weak, low-affinity antibodies.
Optimizing incubation conditions ensures reliable and reproducible western blot results.
A secondary antibody conjugate directed against the host species of the primary antibody is applied. An exception are biotin-labelled primary antibodies, which are detected with streptavidin or avidin conjugated to a reporter molecule.
The secondary antibody carries alkaline phosphatase (AP), which hydrolyses a chromogenic substrate such as BCIP/NBT. The reaction produces a visible, insoluble colored precipitate that builds up directly on the membrane. The developing reaction can be stopped at the exact moment the desired intensity is reached—simply by rinsing the membrane with water or a mild acid. It is less sensitive than chemiluminescence, but its broad dynamic range and permanent signal make it ideal for quick qualitative checks or for archival documentation.
The secondary antibody is conjugated to horseradish peroxidase (HRP). When a luminol‑based substrate is added, HRP catalyzes a rapid chemiluminescent reaction. The light is captured on photographic film or by a chemiluminescence imager. ECL offers the highest sensitivity of the three formats, capable of detecting sub‑picogram protein amounts. Different substrate formulations are available that cover a broad range of sensitivity.
The main disadvantage is a narrow linear range - often only one to two orders of magnitude - so the assay must be carefully titrated and exposure times optimized. Because the signal decays quickly, timing of image acquisition is critical.
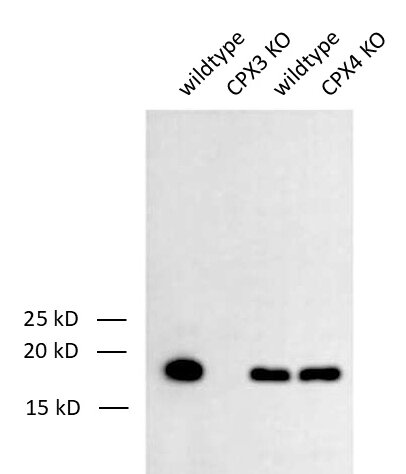
Figure 3: Detection of Complexin3 (CPX3) in WT and KO mouse brain lysates with mouse anti- Complexin3 antibody (cat. no. 122 311, dilution 1:1000). ECL has been used for detection.
Figure 3: Detection of Complexin3 (CPX3) in WT and KO mouse brain lysates with mouse anti- Complexin3 antibody (cat. no. 122 311, dilution 1:1000). ECL has been used for detection.
Fluorophore‑conjugated secondary antibodies emit light after excitation at a specific wavelength. The emitted fluorescence is recorded with a specialized imaging system. Fluorescent detection gives a wide linear dynamic range (typically three to four orders of magnitude), which is valuable for accurate protein quantification. Additionally, because fluorophores can be chosen with non‑overlapping spectra, several proteins can be probed simultaneously on the same membrane—a powerful multiplexing capability. The method is moderately sensitive—generally a little less than the best ECL substrates—but it does not suffer from rapid signal decay.
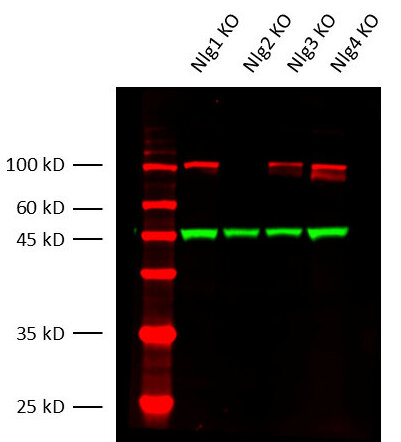
Secondary antibodies can cross-react with endogenous immunoglobulins in tissue (e.g. spleen), leading to non-specific staining. To minimize this:
SYSY Antibodies recommends performing a control without a primary antibody to control for non-specific binding of the secondary reagent. This ensures that any observed signal is due to the specific binding of the primary antibody and not to artifacts of the detection system.