
Antibody specificity is the property of an immunoglobulin that enables it to bind exclusively (or with markedly higher affinity) to a particular antigen while exhibiting minimal or no binding to other, structurally unrelated epitopes.
Even the best antibody will have weak cross-reactivity to off-targets. Increasing the protein amount loaded to the gel will lead to high protein concentrations on the membrane that can create a dense adsorptive surface that promotes nonspecific IgG binding. Increasing the primary antibody concentration even increases this undesired effect. Similarly, highly sensitive detection methods, such as enhanced chemiluminescence, can reveal nonspecific interactions.
SDS-PAGE can resolve only about 50–100 distinct bands. If the target antigen represents less than ~0.2% of total protein, detection may be challenging. For example, Synaptobrevin/VAMP co-migrates with histones in crude homogenates, making it difficult to visualize. Attempts to enhance signal intensity may inadvertently produce artificial bands.
Sometime incomplete or uneven transfer of proteins to the blotting membrane is observed.
A variety of blocking agents are described in the literature, including detergents and protein-based blockers. Inadequate or suboptimal blocking may increase nonspecific binding.
Proteolysis can occur, especially if samples are not freshly prepared, are stored for extended periods, or are subjected to fractionation after homogenization. Proteolytic fragments typically appear as additional bands of lower molecular weight than the full-length protein.
Several bands are not always the result of suboptimal experimental conditions or cross reactivity. Many proteins have several isoforms, and an antibody may detect more than one of them.
Alternative splicing produces mRNA isoforms that encode protein isoforms that often differ in amino‑acid length, domain composition or post‑translational modification (PTM) sites. When the protein mixture is run on an SDS‑PAGE each isoform can migrate to a different apparent molecular weight (MW). Probing with an antibody directed against a shared epitope can give rise to multiple bands.
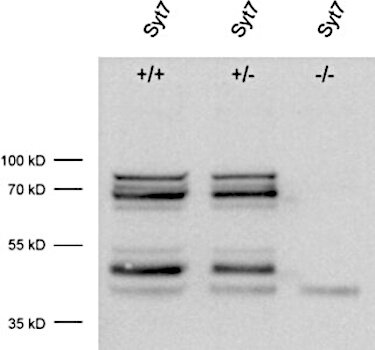
Figure 1: Detection of Synaptotagmin7 in WT and KO mouse brain lysate with rabbit anti Synaptotagmin7 antibody (cat. no. 105 173, dilution 1:1000).
Several splice variants are detected by this antibody, that disappear in the KO control proving the specificity of the signals detected.
The low molecular weight band present in all three samples has to be considered as unspecific.
Figure 1: Detection of Synaptotagmin7 in WT and KO mouse brain lysate with rabbit anti Synaptotagmin7 antibody (cat. no. 105 173, dilution 1:1000).
Several splice variants are detected by this antibody, that disappear in the KO control proving the specificity of the signals detected.
The low molecular weight band present in all three samples has to be considered as unspecific.
In enhanced‑chemiluminescence (ECL) detection, horseradish peroxidase (HRP) catalyzes a rapid luminescent reaction as soon as the substrate cocktail is added to the membrane. If the target protein is abundant and the primary‑/secondary‑antibody pair binds with high affinity, the reaction can generate a burst of light that exhausts the substrate before the camera or imaging system is even triggered. The resulting image contains “burned” or oversaturated bands that obscure quantitative information.
By simultaneously lowering the concentrations of both primary and secondary antibodies, and by minimizing the time between substrate addition and image capture, the flash of light is spread over a longer period, preventing burned bands while preserving sensitivity.
General fluorescent background can be due to Coomassie or bromphenole blue (BPB) contamination on the membrane.
The speckled, smeary background that frequently obscures western blot membranes is most often due to keratin contamination, a problem that can arise from both the samples being analyzed, and the reagents used throughout the procedure. Keratins are a family of high‑molecular‑weight, highly fibrous structural proteins that are abundant in skin, hair, nails and the outer layers of the laboratory environment itself. Because they are inherently “sticky,” even trace amounts that enter a blot can produce a diffuse, grainy haze or a series of faint, smeared bands that span a wide molecular‑weight range.